
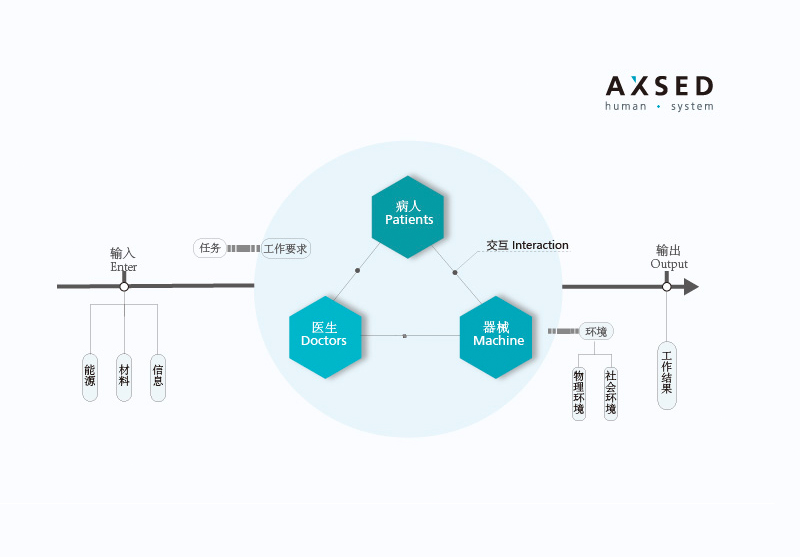
The compliance and agile parallelism is of great importance throughout the development process of medical devices. It includes the definition of product strategy, preliminary investigation of law and regulation, patent layout, usability engineering, user research innovation, the establishment of compliant and normative technique criterion, and how to meet the requirements of top-level design like quality management system and documentation, risk management and quality assurance, etc. All of these are the complex and systematically concurrent process. The quality assurance of products is on the basis of design. Its key is in the early-stage R&D of concurrent project.--AXSED agile compliance and quality system is willing to contribute to your success.
- Discover More|
公司:苏州系能可用性工程设计有限公司 地址:苏州市吴中区东吴北路8号国裕大厦807 苏州市高新区锦峰路8号2号楼411 电话:0512-66798628/13914031785 邮箱:wxf@axsed.com 系能岸刻医疗设计公司专注于医疗器械产品设计开发,涵盖医疗设备外观设计、防疫护理产品设计、手持产品设计 |